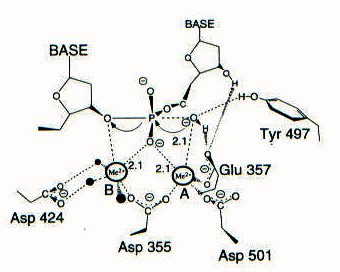
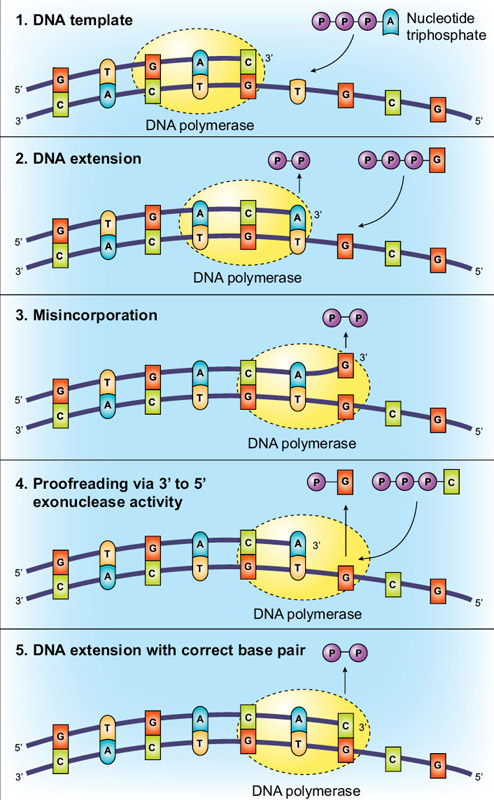
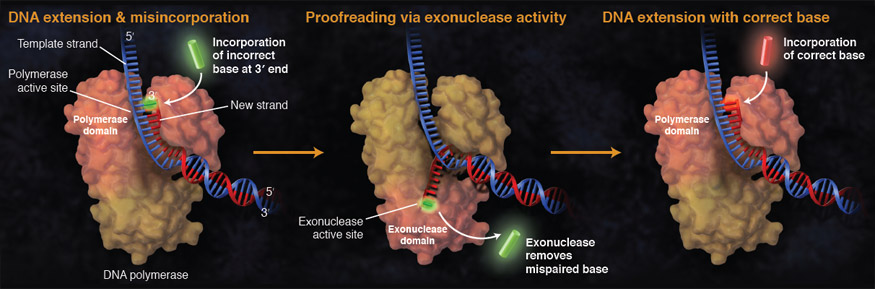
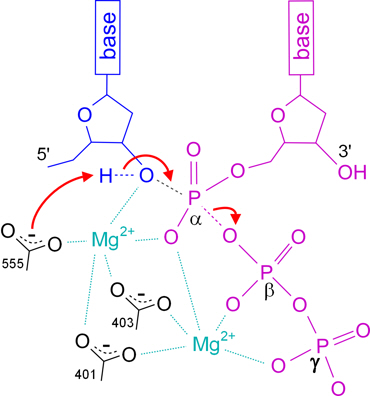
Divalent ions attenuate DNA synthesis by human DNA polymerase α by changing the structure of the template/primer or by perturbing the polymerase reaction. - Abstract - Europe PMC
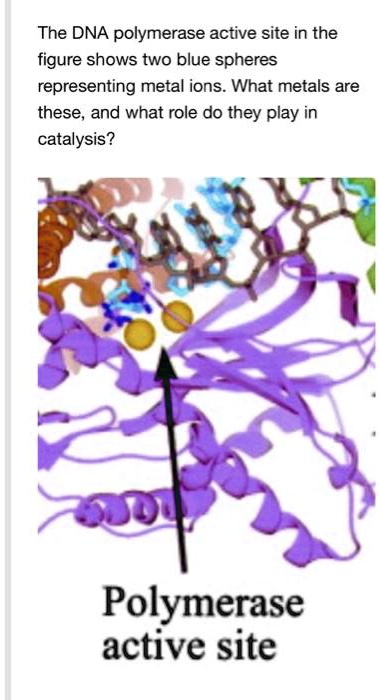
SOLVED: The DNA polymerase active site in the figure shows two blue spheres representing metal ions. What metals are these, and what role do they play in catalysis? Polymerase active site

Highly Organized but Pliant Active Site of DNA Polymerase β: Compensatory Mechanisms in Mutant Enzymes Revealed by Dynamics Simulations and Energy Analyses: Biophysical Journal
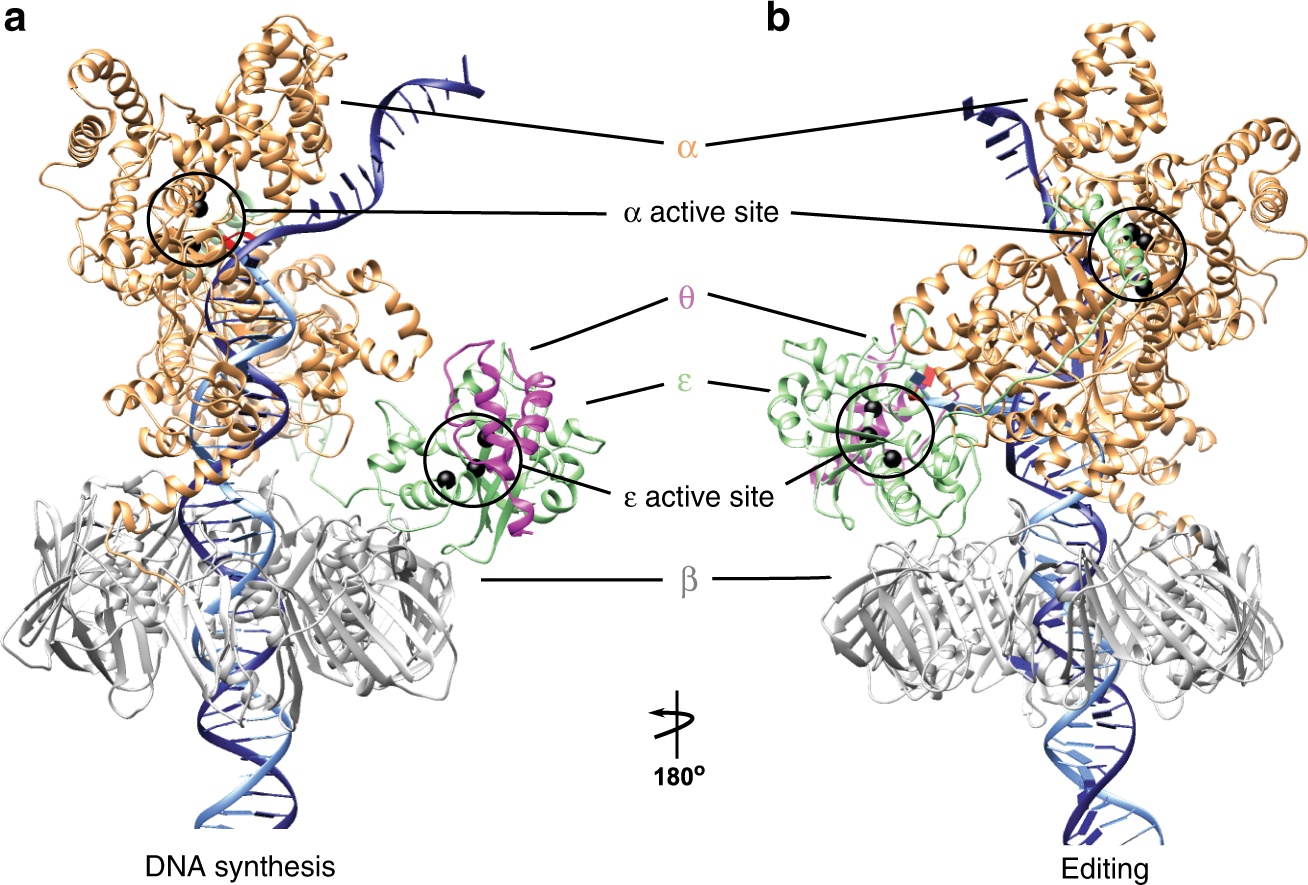
Polymerization and editing modes of a high-fidelity DNA polymerase are linked by a well-defined path | Nature Communications

Critical Role of Magnesium Ions in DNA Polymerase β's Closing and Active Site Assembly | Journal of the American Chemical Society
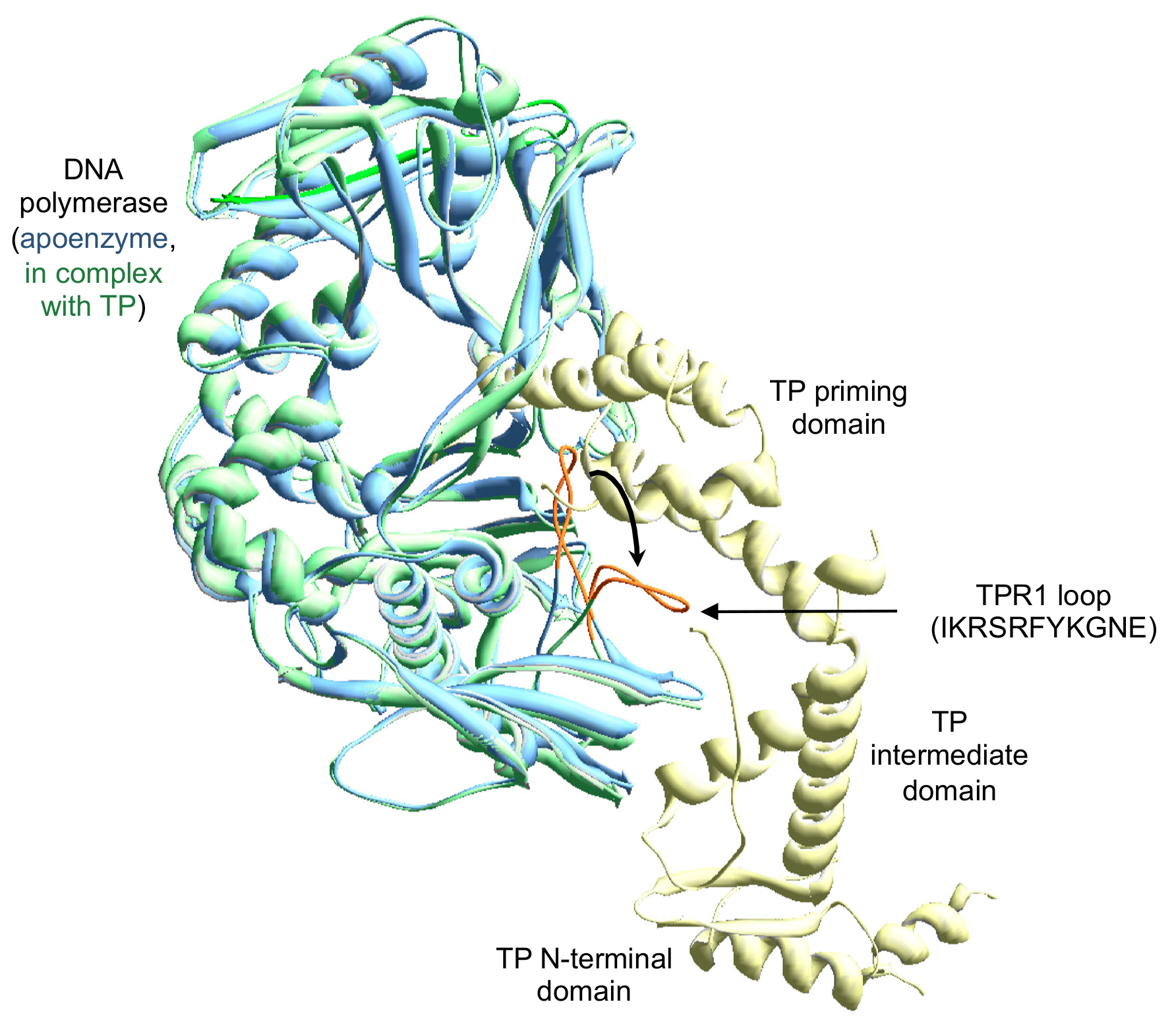
Biomolecules | Free Full-Text | The Loop of the TPR1 Subdomain of Phi29 DNA Polymerase Plays a Pivotal Role in Primer-Terminus Stabilization at the Polymerization Active Site
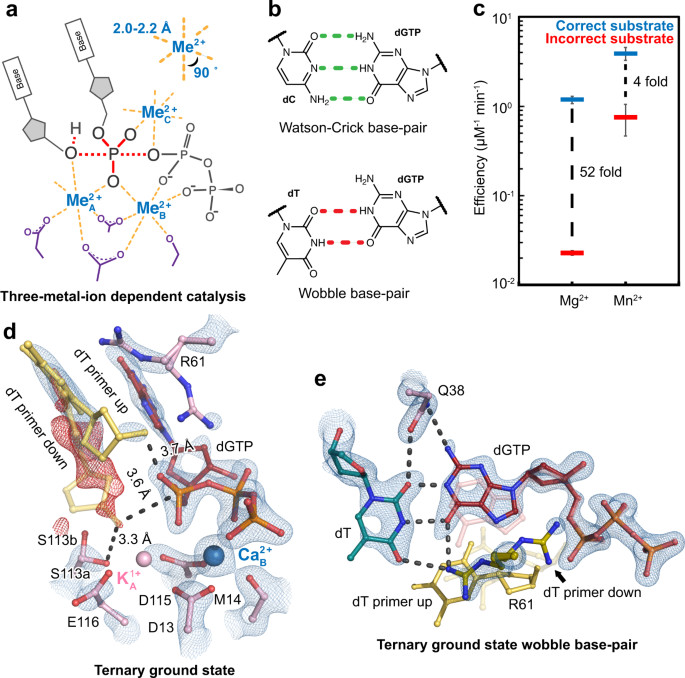
In crystallo observation of three metal ion promoted DNA polymerase misincorporation | Nature Communications

Close-up view of the polymerase active site region.The fingers, palm,... | Download Scientific Diagram
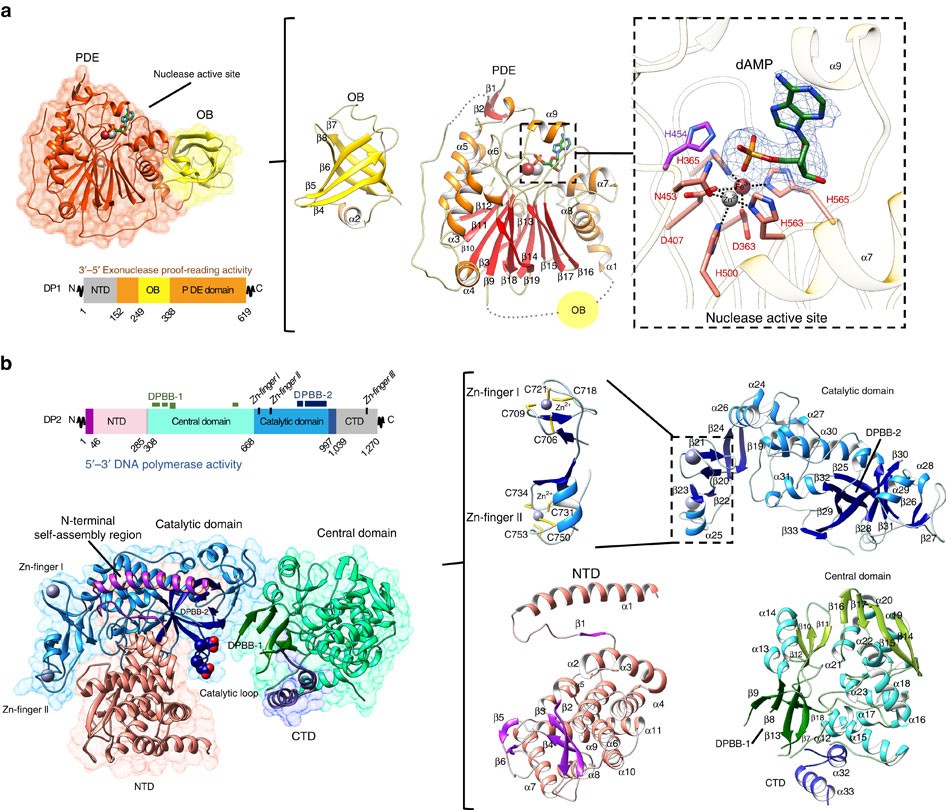
Shared active site architecture between archaeal PolD and multi-subunit RNA polymerases revealed by X-ray crystallography | Nature Communications

Insight into the mechanism of DNA synthesis by human terminal deoxynucleotidyltransferase | Life Science Alliance

The Structural Basis for Processing of Unnatural Base Pairs by DNA Polymerases - Marx - 2020 - Chemistry – A European Journal - Wiley Online Library

Critical Role of Magnesium Ions in DNA Polymerase β's Closing and Active Site Assembly | Journal of the American Chemical Society